| Names | |
|---|---|
| IUPAC name nickel;N-[(Z)-3-nitrosobut-2-en-2-yl]hydroxylamine | |
| Other names | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
| EC Number | |
PubChemCID | |
| |
| |
| Properties | |
| C8H14N4NiO4 | |
| Molar mass | 288.917 g·mol−1 |
| Appearance | red solid |
| Density | 1.698 g/cm3 |
| Hazards | |
| GHS pictograms | |
| GHS Signal word | Warning |
| H315, H317, H319, H335, H351 | |
| P201, P202, P261, P264, P271, P272, P280, P281, P302+352, P304+340, P305+351+338, P308+313, P312, P321, P332+313, P333+313, P337+313, P362, P363, P403+233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
- Structure Of Ni Dmg Complex 4
- Structure Of Ni(dmg)2 Complex
- Structure Of Ni Dmg Complex System
- Structure Of Ni Dmg Complex 1
- Write The Structure Of Ni Dmg Complex
Nickel-dimethylglyoxime complex (abbreviated as Ni(II)(DMG)2) modified carbon paste and graphite electrodes were prepared by mixing Ni(II)(DMG)2 with graphite paste, and coating Ni(II)(DMG)2 to the graphite surface. It is necessary to cycle the electrode potential to a high value (e.g. 0.8V versus SCE) for the preparation of the modified electrodes. The complex is distinctively colored and insoluble leading to its use as a chelating agent in the gravimetric analysis of nickel. The use of dimethylglyoxime as a reagent to detect nickel was reported by L. Chugaev in 1905.
- Jan 09, 2008 Structure of Ni(dmgH)2.As an analytical reagent, dmgH2 is often used as a solution in ethanol. It is the conjugate base, not dmgH2 itself, that forms the complexes. Furthermore, a pair of dmgH- ligands are joined through hydrogen bonds to give a macrocyclic ligand.
- Complex shows semiconductive behavior at high pressures. A crystal structure of Ni(dmg)2 and Pt(dmg)2 is orthorhombic, space group Ibam 5, 6. Both complexes crystallize in linear metal chain columnar structures. Powder x-ray diffraction of M(dmg)2 (M = Ni and Pt) has been studied with synchrotron radiation at high pressures 2, 4.
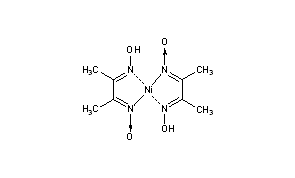
Nickel bis(dimethylglyoximate) is the coordination complex with the formula Ni[ONC(CH3)C(CH3)NOH]2. The compound is a bright red solid. It achieved prominence for its use in the qualitative analysis of nickel.[1]
Structure[edit]
Nickel(II) is square planar.[2] It is surrounded by two equivalents of the conjugate base (dmgH−) of dimethylglyoxime (dmgH2). The pair of organic ligands are joined through hydrogen bonds to give a macrocyclic ligand. The complex is distinctively colored and insoluble leading to its use as a chelating agent in the gravimetric analysis of nickel.
The use of dimethylglyoxime as a reagent to detect nickel was reported by L. A. Chugaev in 1905.[3]
References[edit]
- ^Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN978-0-08-037941-8.
- ^Donald E. Williams, Gabriele Wohlauer, R. E. Rundle (1959). 'Crystal Structures of Nickel and Palladium Dimethylglyoximes'. J. Am. Chem. Soc. 81: 755–756. doi:10.1021/ja01512a066.CS1 maint: uses authors parameter (link)
- ^Tschugaeff, Lev (1905). 'Über ein neues, empfindliches Reagens auf Nickel' [About a new, sensitive reagent on nickel] (PDF). Berichte der deutschen chemischen Gesellschaft (in German). 38 (3): 2520–2522. doi:10.1002/cber.19050380317.
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Nickel_bis(dimethylglyoximate)&oldid=935161854'
1) Amongst Ni(CO)4, [Ni(CN)4]2- and NiCl42-:
(IIT JEE 1991)
a) Ni(CO)4 and NiCl42- are diamagnetic; and [Ni(CN)4] 2- is paramagnetic.
b) [Ni(CN)4]2- and NiCl42- are diamagnetic; and Ni(CO)4 is paramagnetic.
c) Ni(CO)4 and [Ni(CN)4]2- are diamagnetic; and NiCl42- is paramagnetic.
d) Ni(CO)4 is diamagnetic; [Ni(CN)4]2- and NiCl42- are paramagnetic.
Logic & Solution:
Ni(CO)4 = Ni + 4CO
* The valence shell electronic configuration of ground state Ni atom is 3d8 4s2.
* All of these 10 electrons are pushed into 3d orbitals and get paired up when strong field CO ligands approach Ni atom. The empty 4s and three 4p orbitals undergo sp3 hybridization and form bonds with CO ligands to give Ni(CO)4. Thus Ni(CO)4 is diamagnetic.
KEY POINTS:
Ni(CO)4 Hybridization:sp3
Ni(CO)4 Shape & Structure (geometry): Tetrahedral
Ni(CO)4 Magnetic nature: Diamagnetic (low spin)
[Ni(CN)4]2- = Ni2+ + 4CN-
* In [Ni(CN)4]2-, there is Ni2+ ion for which the electronic configuration in the valence shell is 3d8 4s0.
* In presence of strong field CN- ions, all the electrons are paired up. The empty 4d, 3s and two 4p orbitals undergo dsp2 hybridization to make bonds with CN- ligands in square planar geometry. Thus [Ni(CN)4]2- is diamagnetic.
KEY POINTS:
[Ni(CN)4]2- Hybridization:dsp2
[Ni(CN)4]2- Shape: Square planar
[Ni(CN)4]2- Magnetic nature: Diamagnetic (low spin)
NiCl42-= Ni2+ + 4Cl-
* Again in NiCl42-, there is Ni2+ ion, However, in presence of weak field Cl- ligands, NO pairing of d-electrons occurs. Therefore, Ni2+ undergoes sp3 hybridization to make bonds with Cl- ligands in tetrahedral geometry. As there are unpaired electrons in the d-orbitals, NiCl42- is paramagnetic and is referred to as a high spin complex.
KEY POINTS:
[NiCl4]2- Hybridization:sp3
[NiCl4]2- Shape & Structure: Tetrahedral
[NiCl4]2- Magnetic nature: Paramagnetic (low spin)
Conclusion:
The correct option is 'c'.
Related questions
2) Among the following the lowest degree of paramagnetism per mole of the compound at 298 K will be shown by:
(IIT JEE 1988)
a) MnSO4.4H2O
b) CuSO4.5H2O
c) FeSO4.6H2O
d) NiSO4.6H2O
Logic & solution:
* In presence of water, which is a weak field ligand, the configurations of metal ions in hydrated compounds reflect those in isolated gaseous ions i.e., no pairing of electrons is possible as the interaction with water molecules is weak.
* All the metal ions in the above compounds are divalent and their outer shell electronic configurations are shown below.
* The paramagnetic nature of a compound is proportional to the number of unpaired electrons in it. Mn2+ ion has more number of unpaired electrons. Hence MnSO4.4H2O shows greater paramagnetic nature.
Whereas there is only one unpaired electron in Cu2+ and hence CuSO4.5H2O shows lowest degree of paramagnetism.
Conclusion:
Structure Of Ni Dmg Complex 4
Correct option is: 'b'.
3) Among the following ions which one has the highest paramagnetism?
(IIT JEE 1993)
a) [Cr(H2O)6]3+
b) [Fe(H2O)6]2+
c) [Cu(H2O)6]2+
d) [Zn(H2O)6]2+
Structure Of Ni(dmg)2 Complex
Logic & solution:
* As mentioned in previous question, the electronic configuration of metal ions is not much affected by weak field ligand water. Hence the electronic configurations of metal ions coordinated to water are same as in isolated ions.
* The outer shell electronic configurations of metal ions in the above complexes are shown below.
* Fe2+ ion has more number of unpaired electrons. Hence [Fe(H2O)6]2+ is more paramagnetic.
Note: The charge on metal ions is equal to the charge on the complex since water is a neutral ligand.
Conclusion:
Correct option is: 'b'.
4) Transition metals show paramagnetic behavior. This is because of their :
(AFMC 2009)
A) high lattice energy
B) variable oxidation state
Structure Of Ni Dmg Complex System
C) characteristic configuration
D) unpaired electrons
Conclusion:
Correct option is: D
Structure Of Ni Dmg Complex 1
Homework
1) How do you calculate the magnetic moment of ions of transition elements?
2) What are the strong field and weak field ligands?
3) Why Ti(SO4)2 is diamagnetic?
4) What is the hybridization & structure of [CoCl4]2-? What is the magnetic nature of this compound?
Write The Structure Of Ni Dmg Complex
(new) Click here to see 3d Interactive Solved Question paper
